Recently, Cygnus Bio and Alamar Biosciences jointly completed the performance verification of the NULISAseq™ CNS Disease Panel 120. The results show that the independently developed GP1000 (P1000) Sequencing System by Cygnus Bio has comprehensively matched international mainstream platforms in data quality for proteomics detection, providing researchers with more new options.
1. NULISA™ Technology: High-Throughput Sequencing for Protein Quantification
NULISA™ (NUcleic acid-Linked Immuno-Sandwich Assay) is a new type of ultra-sensitive, high-throughput immunoassay technology developed by Alamar Biosciences. With its unique "immune complex" framework design and fully automated workflow, it achieves 10,000-fold noise reduction and exponential signal amplification while ensuring specificity, providing extremely high detection signal-to-noise ratio and sensitivity: the limit of detection (LOD) can reach sub-femtogram level (for example, the LOD of IL-13 is 0.2 fg/mL), and the limit of quantitation (LOQ) can reach femtogram level (fg/mL).
2. Verification Methods and Quality Control Indicators
A total of 86 human plasma samples and cerebrospinal fluid samples were used in this verification. The selected NULISAseq™ CNS Disease Panel 120 includes 127 proteins related to central nervous system (CNS) diseases. All samples were subjected to library construction in strict accordance with the standard NULISAseq™ process, and each library was split into two parts, which were run simultaneously on the Cygnus Bio GP1000 (P1000) Sequencing System and the validated Sequencing System of Alamar Biosciences for horizontal comparison.
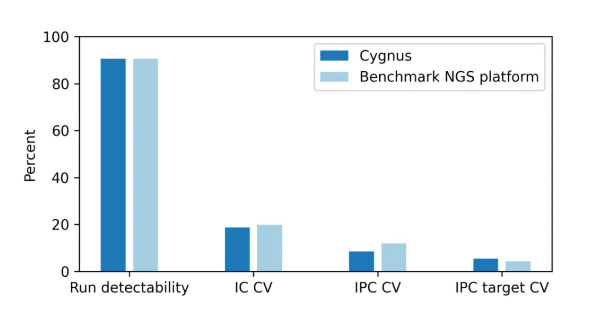
Four key quality control indicators were focused on:
Sequencing results show that in the detection of NULISAseq™ libraries, the key quality control indicators of the Cygnus Bio GP1000 (P1000) Sequencing System are highly consistent with those of Alamar Biosciences' validated Sequencing System. Its detection capability, signal stability and batch reproducibility all reach the same level, fully ensuring data reliability. More notably, the GP1000 (P1000) performed better in the two indicators of IC CV and IPC CV, with higher immunocapture efficiency and more stable internal reference calibration, further improving detection accuracy.
3. Core Results: High Correlation and Consistency
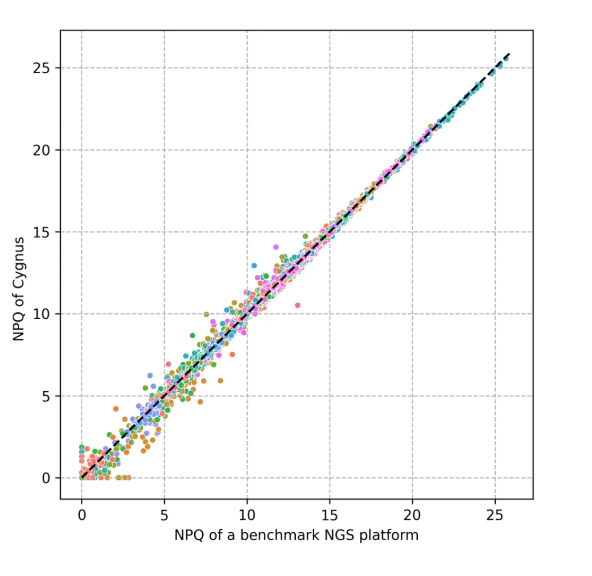
NULISA™ technology uses the NULISA Protein Quantification (NPQ) value to characterize the abundance of each protein. The NPQ value is based on the number of DNA tags corresponding to the target protein, and the number of these tags is directly proportional to the abundance of the target protein. The verification results show that there is an extremely high correlation (R²=0.9949) between the NPQ values measured by the Cygnus Bio GP1000 (P1000) Sequencing System and Alamar Biosciences' validated Sequencing System.
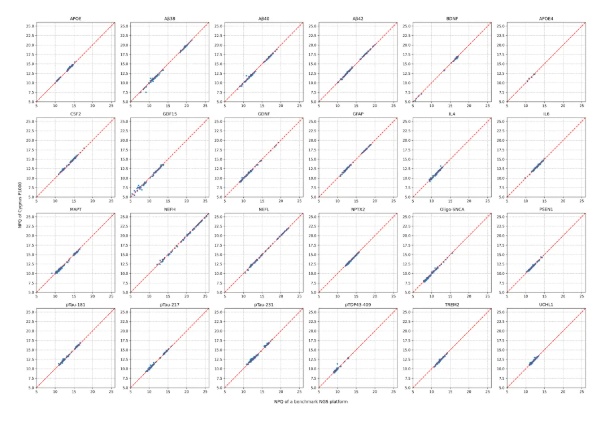
In addition, to further verify the specificity performance of the GP1000 (P1000) Sequencing System, we also paid special attention to 24 very important proteins in the Panel, including low-abundance targets such as pTau217, pTau181, pTau231, GFAP, and NfL. The results show that for these key proteins, the NPQ values measured by the two platforms still maintain a high degree of consistency, which once again confirms the reliability of the GP1000 (P1000) Sequencing System in the analysis of specific biomarkers.
Conclusion
This joint verification fully and strongly confirms that the Cygnus Bio GP1000 (P1000) Sequencing System has excellent performance, can stably and efficiently adapt to Alamar Biosciences' NULISAseq™ proteomics technology, and can output high-credibility data equivalent to that of mainstream platforms.





